

biotech Fungal infections are a global threat that is becoming increasingly severe World Health Organization says. Biosergen, a clinical stage biotech company, is developing a cure, which also is reducing resistance development of the fungus.
Biosergen’s lead compound BSG005 is currently being tested in humans in a clinical phase 1 trial in Australia. The purpose of the trial is to test tolerability, safety, what the body does to the drug and what the drug does to the body. The first part of the trial, the single ascending dose is completed. This part consisted of four groups of six humans, where each group received a higher dose of the drug. The study has advanced to the second part, the Multi Ascending Dose part. Here four groups of six humans will receive the drug once daily for 7 days and each group will receive a higher dose than the preceding group. The first group have been completed and we have progressed to the second group.
Topline data from the phase 1 trial is expected in Q2 this year and we expect begin testing the drug in patients later this year.
Fungal strain growth in a laboratory
BSG005 is aimed for treating Invasive Fungal Infections. Annually there are 1.7 M cases of life threatening invasive fungal infections from just 4 of the most usual fungal strains. Mortality varies with fungal strain and country but is between 20 and 85%.
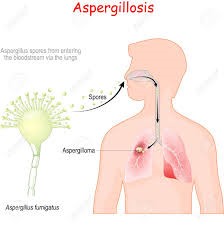
Aspergillosis fungi spores entry into lungs
Fungal infections are a global threat that is becoming increasingly problematic according to WHO. The number of humans diagnosed with Invasive Fungal Infections is rising because more fungal strains resistant to existing treatments are emerging. In addition to this more people live with a reduced immune system due to an aging population, more cancer treatments, more transplants, and people with AIDS.
WHO says that fungal infections and their increasing resistance to treatment is a growing risk. The historic lack of focus on the danger is the reason to large gaps in knowledge, as well as a lack of surveillance, treatments, and diagnostics. Biosergen is addressing the problem with increasing resistance to current drugs and develop a new drug with higher potency and effect and which does not have adverse impact on the kidneys and the liver.
Most existing treatments against fungal infections are inhibiting the growth of the fungal strain. Only a few of the treatments available are killing the fungal strain and thereby preventing resistance from occurring.
Biosergen’s lead development compound BSG005 is a compound that kills the fungal strain (is fungicidal). BSG005 is a novel anti-fungal molecule, a polyene macrolide, a group of molecules known for their broad anti-fungal and fungicidal effect, including on resistant and difficult-to-treat fungal strains.
Fungi growth in the lungs
There are other polyene macrolides to treat fungal infections, but the main shortcoming of other polyenes are their severe side effects. They are toxic to the kidney and liver and people being treated with these drugs are often taken off the treatment again due to the damage on kidney and liver and returning to treatment with drugs that are not adequate to cure the infection. Due to its broad anti-fungal and fungicidal effect BSG005 and that it is the only polyene without kidney and liver toxicity, BSG005 can break through the barrier and become a candidate for first line therapy against severe invasive fungal infections.
Invasive Fungal Infection are difficult to diagnose and difficult to treat. Determining what fungal strain is causing the infection can take days and the patient may die before the doctor will know what treatment is the right one. Therefore, it is important to have a treatment, a drug with a broad spectrum even against strains who have developed resistance, and which is not toxic to other vital organs. We have designed such a drug and with the expected success in the clinical testing of the drug BSG005 may have the potential to change the way we treat Invasive Fungal Infections.
Biosergen is listed on First North Growth Market in Stockholm.
Detta är en annons från Biosergen och ska inte tolkas som redaktionellt material från Börsvärlden.
Det här är en sponsrad artikel framtagen i samarbete med Biosergen. Sponsrade inlägg är en del av Börsvärldens annonserbjudande. Om du med anledning av våra sponsrade inlägg önskar att komma i kontakt med oss, vänligen skriv till IR@borsvarlden.com.
LÄS MER